- Home
- Pricing
- About Us
- Partners
- Jobs
- Contact Us
- All in One Data Platform
- Fully Customizable
- Adaptable Widgets
- Process Integration
- AI Integration
- Hardware & Software Integration
- Top Tier Security
- User Friendly Interface
- Professional & Speedy Support
- Data Centers
- Barcode/QR Code System
- articleElectronic Lab Notebook (ELN)
- inventory_2Inventory Management
- scienceLaboratory Information Management Systems (LIMS)
- calculateFormulation Management
- yardAgricultural Research Manager (ARM)
- health_and_safetyQuality Management System (QMS)
- businessBusiness Development
- todayProject Management
- attach_moneyFinancial Management
- descriptionReference Management
- differenceOther Applications
- Biotech and Pharmaceutical Companies
- Contract Research Organizations (CROs)
- Clinical Products/Services for Physician Office
- Agricultural Research Companies
- Build Any SaaS Application in Minutes with AI
- Login
- Free signup
- Request a demo
- Referral Program
- Subscribe to our newsletter
- Documentation
- Blogs
- Resources
- Screenshots
- Downloads
- Terms of Service
- Privacy Policy
Electronic Lab Notebook (ELN)
The All-in-One Configurable Electronic Lab Notebook
Overview
Labii Electronic Lab Notebook (ELN) is a next-generation, cloud-based solution that replaces traditional paper lab notebooks with a digital, flexible, and fully customizable platform. Designed to serve scientists, researchers, and manufacturers across industries, Labii ELN enables detailed experimental documentation, structured data capture, real-time collaboration, and secure record-keeping. With its modular design, Labii ELN can be tailored to support any research workflow—from life sciences to industrial R&D—and adapts as your needs evolve. Whether you’re documenting experiments, tracking results, or managing compliance, Labii puts everything you need in one place.
Purpose-Built ELNs for Every Scientific Workflow
Labii offers a suite of specialized Electronic Lab Notebooks to match the unique needs of different scientific domains and industry sectors.
1) The ELN for Research and Development empowers teams to record experiments with full flexibility, supporting diverse scientific disciplines and protocols.
2) The ELN for Production is optimized for regulated manufacturing workflows, enabling batch recordkeeping, SOP compliance, and traceability.
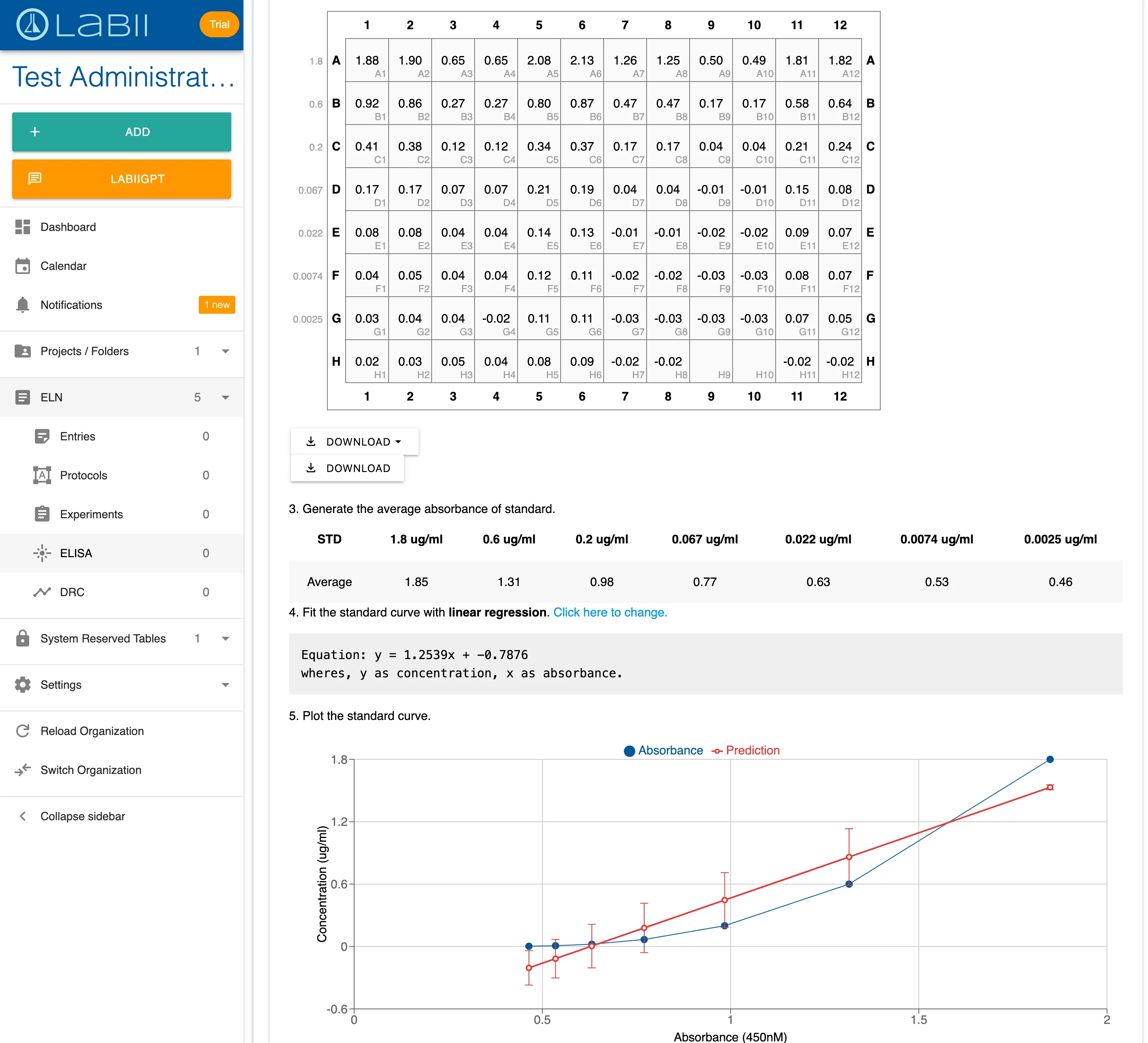
3) The ELN for ELISA simplifies immunoassay data capture with built-in data tables, analysis tools, and visualization.
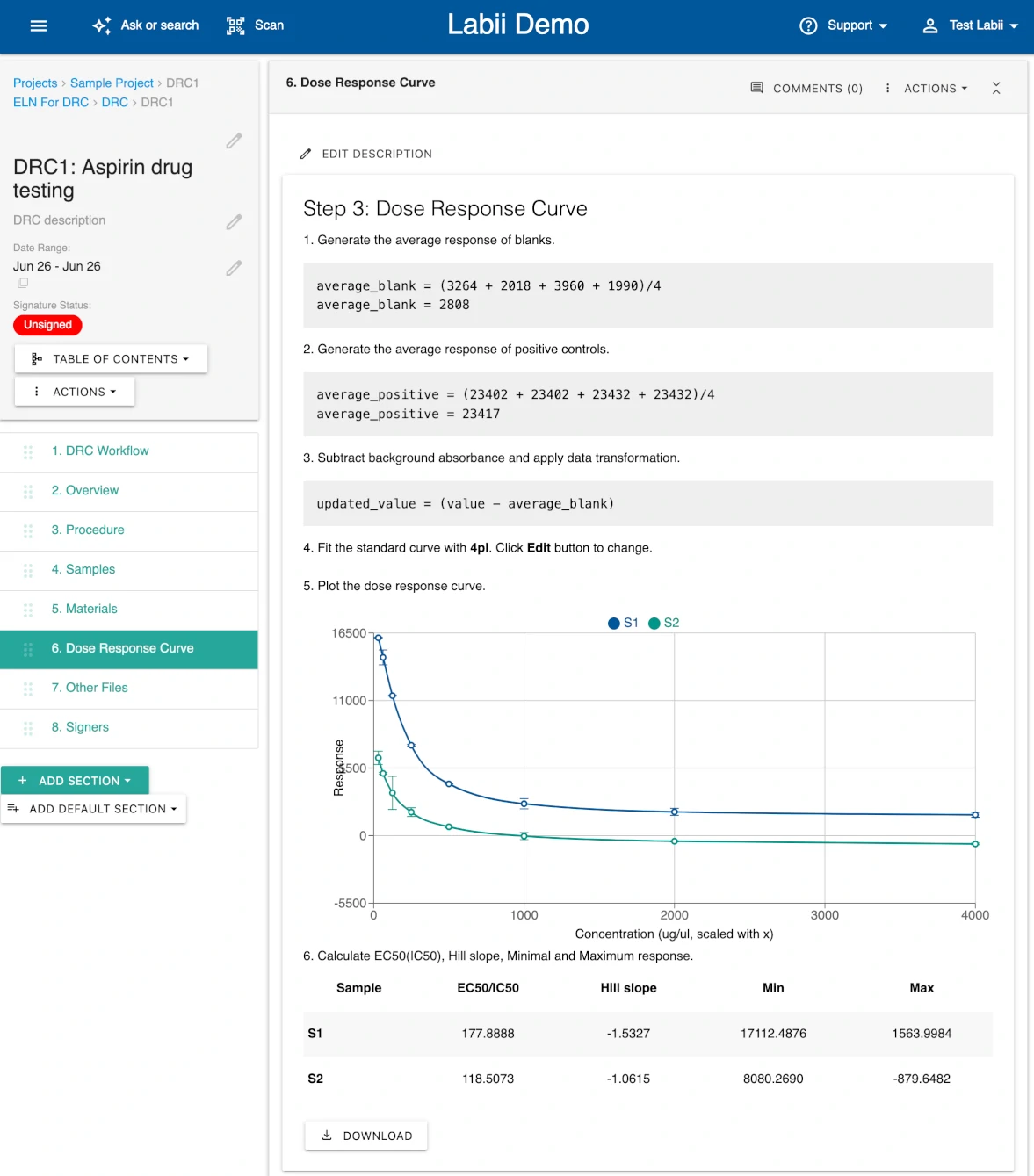
4) The ELN for Dose Response Curve offers integrated curve fitting and automated analysis to streamline pharmacological evaluations.
Each variety can be further customized to fit specific organizational workflows and data structures.
Designed for Compliance with FDA 21 CFR Part 11, GLP, and GxP
Labii ELN meets regulatory requirements for electronic records and signatures, including full compliance with FDA 21 CFR Part 11, GLP, GMP, and GxP standards. The platform includes technical controls such as secure login, user role management, time-stamped audit trails, record versioning, and electronic signature workflows that ensure data integrity and traceability. These features make Labii an ideal ELN for regulated industries like pharmaceuticals, biotechnology, food safety, and diagnostics. Whether you are preparing for audits or operating in a validated environment, Labii helps you stay compliant and inspection-ready at all times.
Fully Customizable Metadata, Layouts, and Workflows
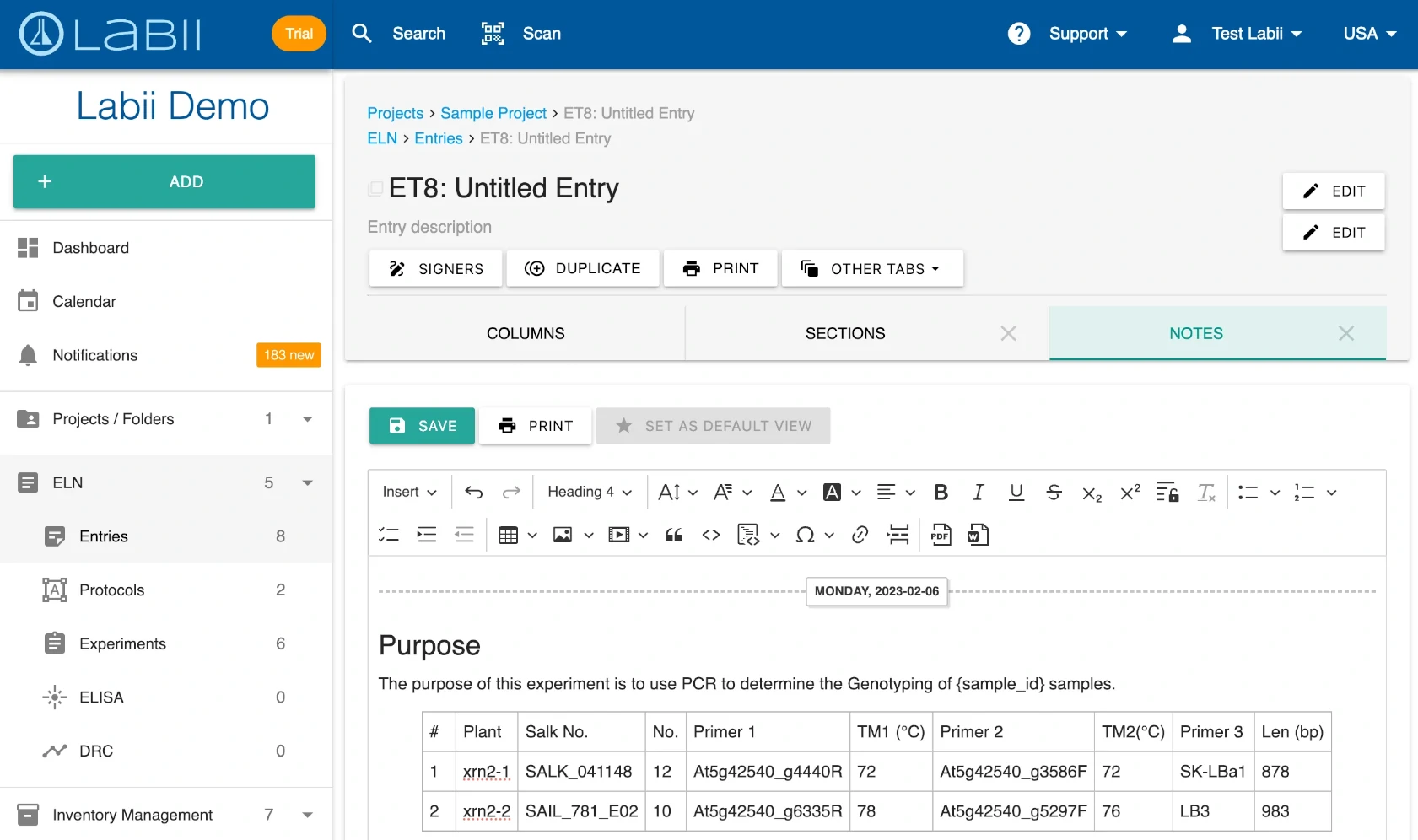
Labii ELN is not a one-size-fits-all solution—it is a fully configurable electronic lab notebook that lets you define your own data structure, workflows, and layouts. You can create and modify metadata columns, sections, widgets, and tables to match your scientific or operational processes. From simple note-taking to complex multi-step experiments, Labii gives you full control over how data is entered, displayed, and reported.
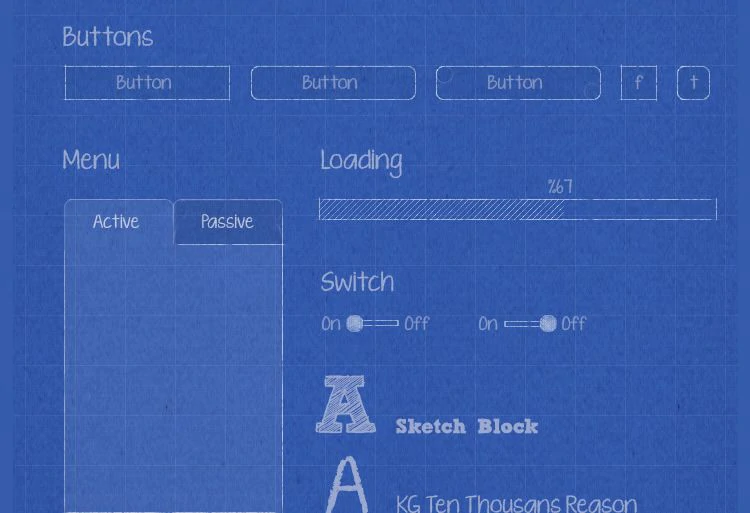
100+ Scientific Widgets for Real-Time Data Analysis and Visualization
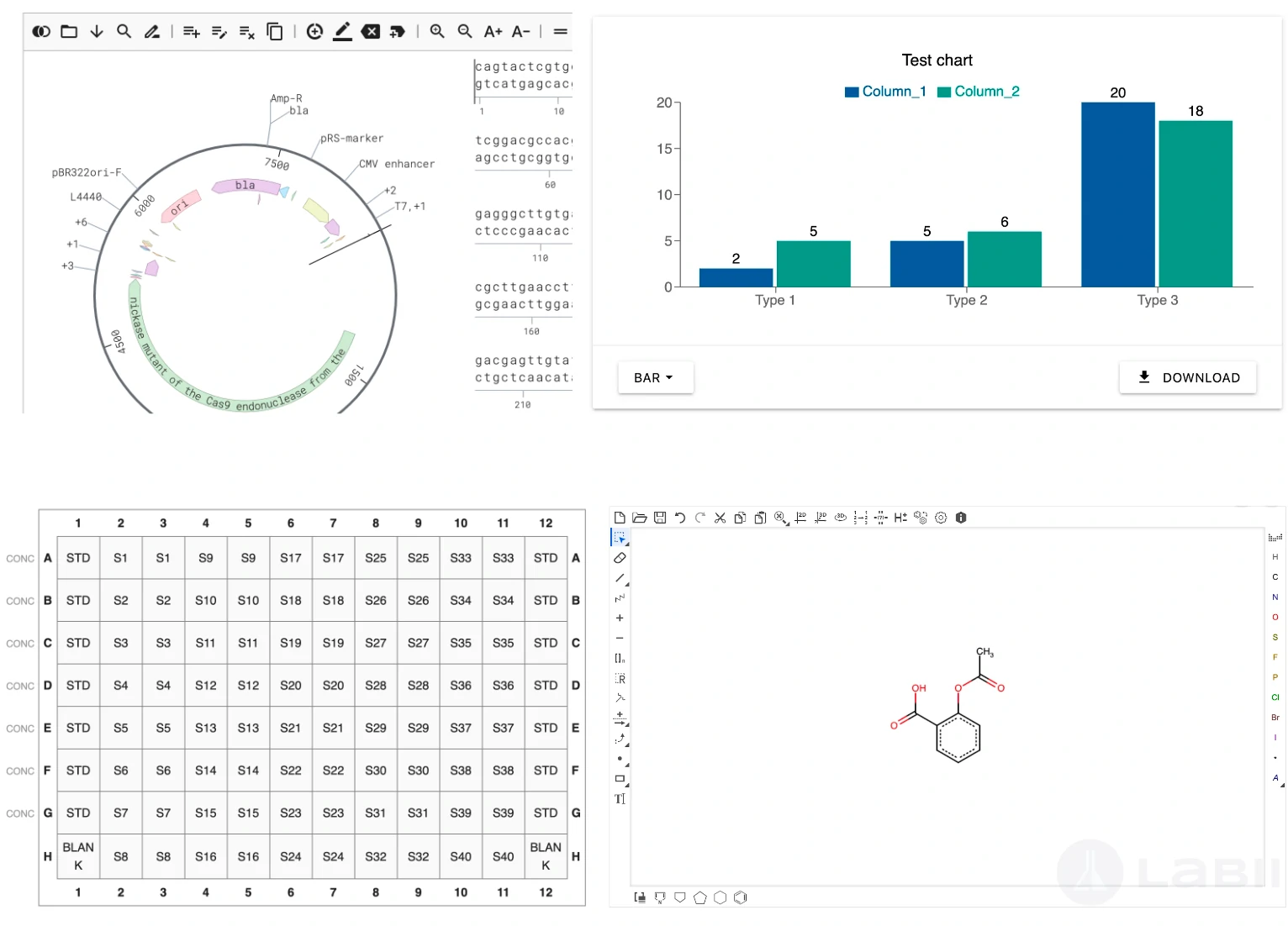
Labii ELN includes a comprehensive library of over 100 scientific data widgets designed to support biological, chemical, and engineering workflows. Widgets enable interactive data visualization, sequence editing, ELISA result plotting, dose-response analysis, image annotation, file viewing, and much more—all within the record interface. These widgets transform Labii from a passive documentation tool into an active data analysis environment, allowing you to visualize, process, and interpret results without switching tools. This level of integration saves time, reduces error, and enhances scientific productivity.
read_moreRead more about widgetsAI-Powered Experiment Notes — Generated in Seconds
Labii’s AI assistant transforms how scientists document their work by automatically generating structured, high-quality experiment notes in seconds. Using machine learning trained on scientific workflows, the AI analyzes your lab activities, procedures, and data entries to draft clear and compliant summaries of what was done. Whether it’s recording steps taken, materials used, or observations made, the AI captures every critical detail—saving hours of manual writing. Ideal for busy research teams and regulated environments, this feature boosts productivity, ensures consistency, and simplifies end-of-day reporting or audit preparation.
Enterprise-Grade Data Security and Privacy Controls
Labii takes data protection seriously, providing enterprise-grade cloud security, end-to-end encryption, user-based access control, and audit logging to safeguard your scientific data. Hosted on secure and compliant infrastructure, Labii ensures redundancy, uptime, and data backup across multiple regions. The platform adheres to industry best practices and supports ISO 27001, GDPR, and HIPAA alignment, making it suitable for organizations that handle sensitive or proprietary information. With Labii, your ELN data is always in secure hands—protected against unauthorized access, data loss, and cyber threats.
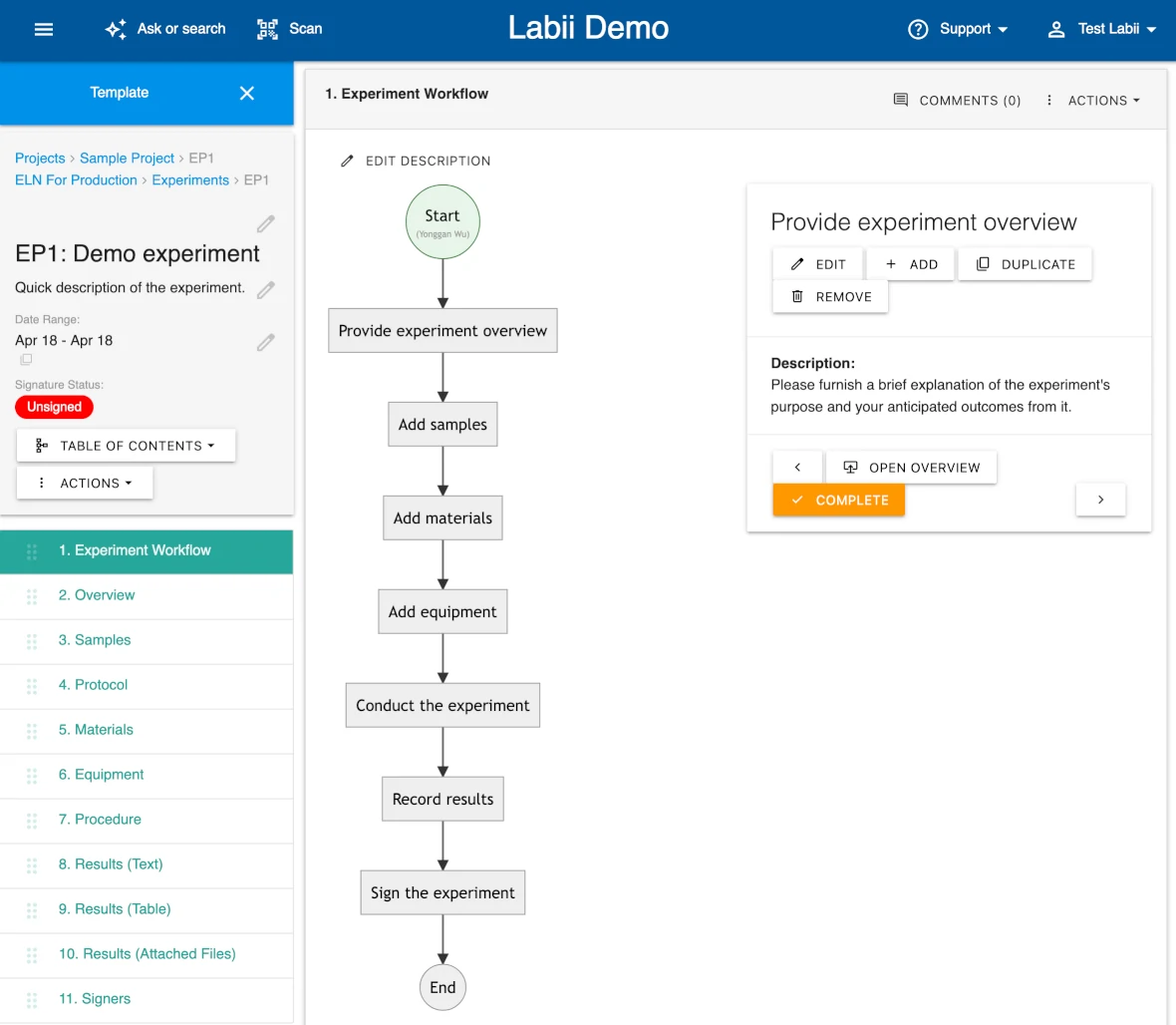
Visual Process Workflows to Standardize Lab Operations
With Labii ELN, you can build and implement automated process workflows that guide users through every step of an experiment or procedure. Define protocols, enforce steps, track progress, and ensure reproducibility across your organization. These workflows help maintain consistency in production-level experiments, simplify training, and reduce human error. Whether used in R&D or manufacturing, Labii’s workflow engine ensures that procedures are followed correctly and that critical data is collected at every step.
read_moreRead more about process integration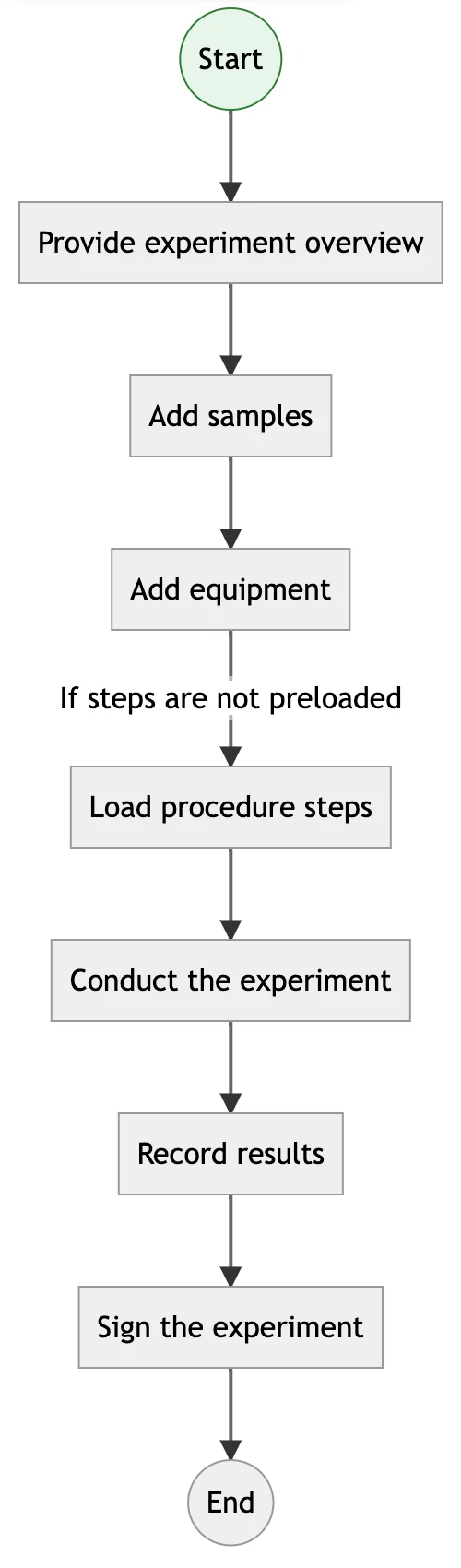
Seamlessly Integrated with Inventory, Samples, and Equipment Management
Labii is more than just an ELN—it’s a complete lab data management platform. The ELN module is deeply integrated with other essential modules like sample tracking, inventory management, equipment scheduling, and storage location mapping. This ensures that your experiment records are directly linked to the physical materials, instruments, and conditions involved. With Labii, you gain full traceability, streamlined operations, and centralized data—all without needing multiple disconnected tools.
Readmore

In the rapidly evolving landscape of scientific research, maintaining accurate and organized records is paramount. Electronic Lab Notebooks (ELNs) have emerged as indispensable tools, offering researchers efficient ways to document experiments, manage data, and integrate various laboratory processes. However, selecting the right ELN can be a daunting task, given the plethora of options available, each with its unique features and capabilities. To assist you in this endeavor, we’ve compiled a list of the top 10 ELNs in 2025, focusing on aspects such as customization and integration with inventory management systems.
The Electronic Laboratory Notebook (ELN) and Laboratory Information Management System (LIMS) market has become an essential part of modern scientific research, streamlining lab workflows and data management for academic, biotechnology, and pharmaceutical labs worldwide. Today, Benchling and Labii stand out as key players in this market, each offering unique approaches to lab data management. Benchling is known for its broad industry adoption, but it comes with a high price tag, making it difficult for many labs and startups to justify the cost. Labii, on the other hand, focuses on affordable, feature-rich solutions that cater to a wide range of labs without the weight of venture capital overhead.
An electronic lab notebook (also known as an electronic laboratory notebook, or ELN for short) is a computer program designed to replace paper lab notebooks that are used for recording experiments. A lab notebook is a type of document that is used by scientists, engineers, and technicians to record research, experiments, and procedures performed in the laboratory. There is a common practice to maintain a lab notebook as a legal document and may even be used as evidence in a court of law. Often referred to in patent prosecution and intellectual property litigation, the lab notebook has a similar function to an inventor's notebook.

As an industry expert and founder of an Electronic Lab Notebook (ELN), I’m closely in touch with over 300 biotech companies/labs and will like to provide a unique comparative analysis of Labii and Benchling. I’ll cover details on Development, IP, Customers, ELN, LIMS, Security, and Price.
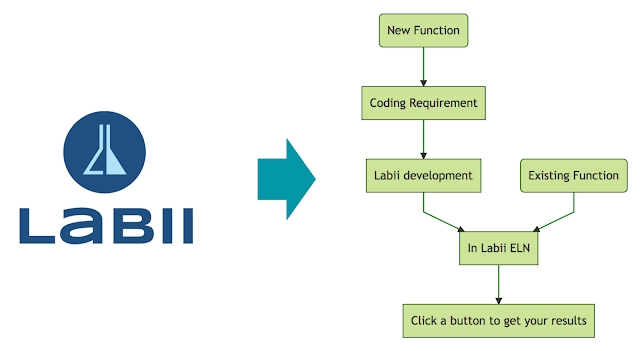
Labii has come up with a solution which is an Electronic Lab Notebook that allows bioinformatics scientists to do their analysis directly in the ELN. This is done via the use of function specific widgets that allows you to get more work done as a bioinformatics scientist.

Oftentimes, when doing demos for prospective customers of our Labii ELN we get the question,”What is a widget?” This scenario is common when computer scientists meet research scientists in the lab. This article will demonstrate the basics of widgets and why Labii implements widgets to Electronic Lab Notebooks.
Ready to Build Your Own Scientific Applications?
See how Labii can transform your research management. Schedule a live demo or start configuring your system today. Whether you’re looking for a complete ELN/LIMS replacement or building your own lab workflows, Labii gives you everything you need to start fast and scale smart.
Refer a company to Labii and earn free months for each seat they sign.