- Home
- Pricing
- About Us
- Partners
- Jobs
- Contact Us
- All in One Data Platform
- Fully Customizable
- Adaptable Widgets
- Process Integration
- AI Integration
- Hardware & Software Integration
- Top Tier Security
- User Friendly Interface
- Professional & Speedy Support
- Data Centers
- Barcode/QR Code System
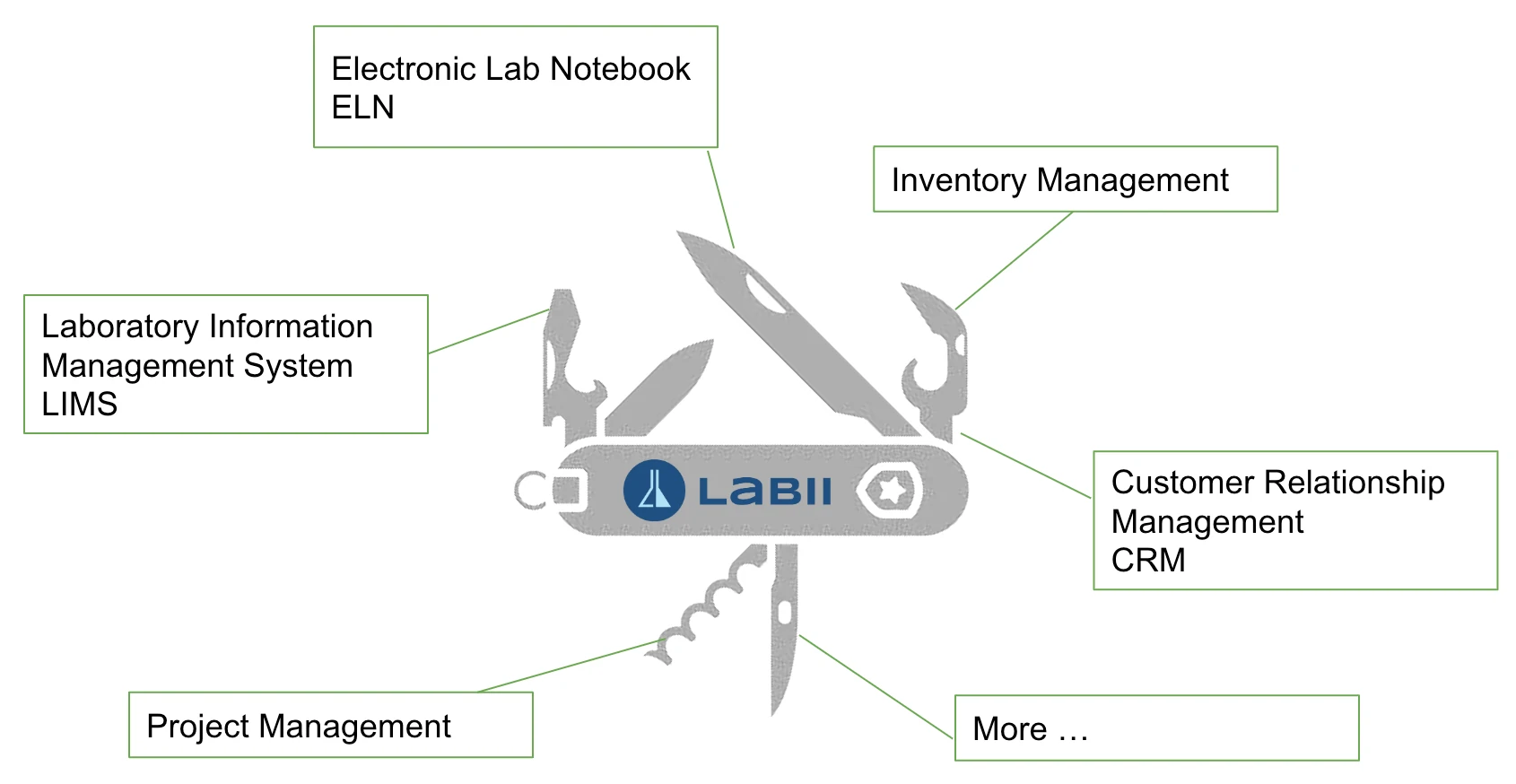
- articleElectronic Lab Notebook (ELN)
- scienceSample Management
- inventory_2Inventory Management
- precision_manufacturingEquipment Management
- biotechLaboratory Information Management System (LIMS)
- timelineChemical Registration
- pest_control_rodentMouse Colony Management
- todayProject Management
- groupsCustomer Relationship Management (CRM)
- attach_moneyPurchase Management
- descriptionReference Management
- co_presentTraining Management
- categoryOntology
- emergencyDiabetes Tracker
- Biotech and Pharmaceutical Companies
- Contract Research Organizations (CROs)
- ELN/LIMS Providers
- Any Organization Needing Data Documentation
- Login
- Free signup
- Request a demo
- Referral Program
- Documentation
- Blogs
- Resources
- Screenshots
- Downloads
- Terms of Service
- Privacy Policy
Electronic Lab Notebook (ELN) for Production
Overview
Labii Electronic Lab Notebook (ELN) for Production is purpose-built to support standardized, repetitive experiments by enabling users to pre-design structured sections that include SOP steps, samples used, equipment involved, expected results, and more. With the ability to configure complex workflows, Labii ensures consistent execution and documentation of production-level protocols. This approach streamlines data entry, improves documentation efficiency, and guarantees that all required details are captured in compliance with defined operational standards.
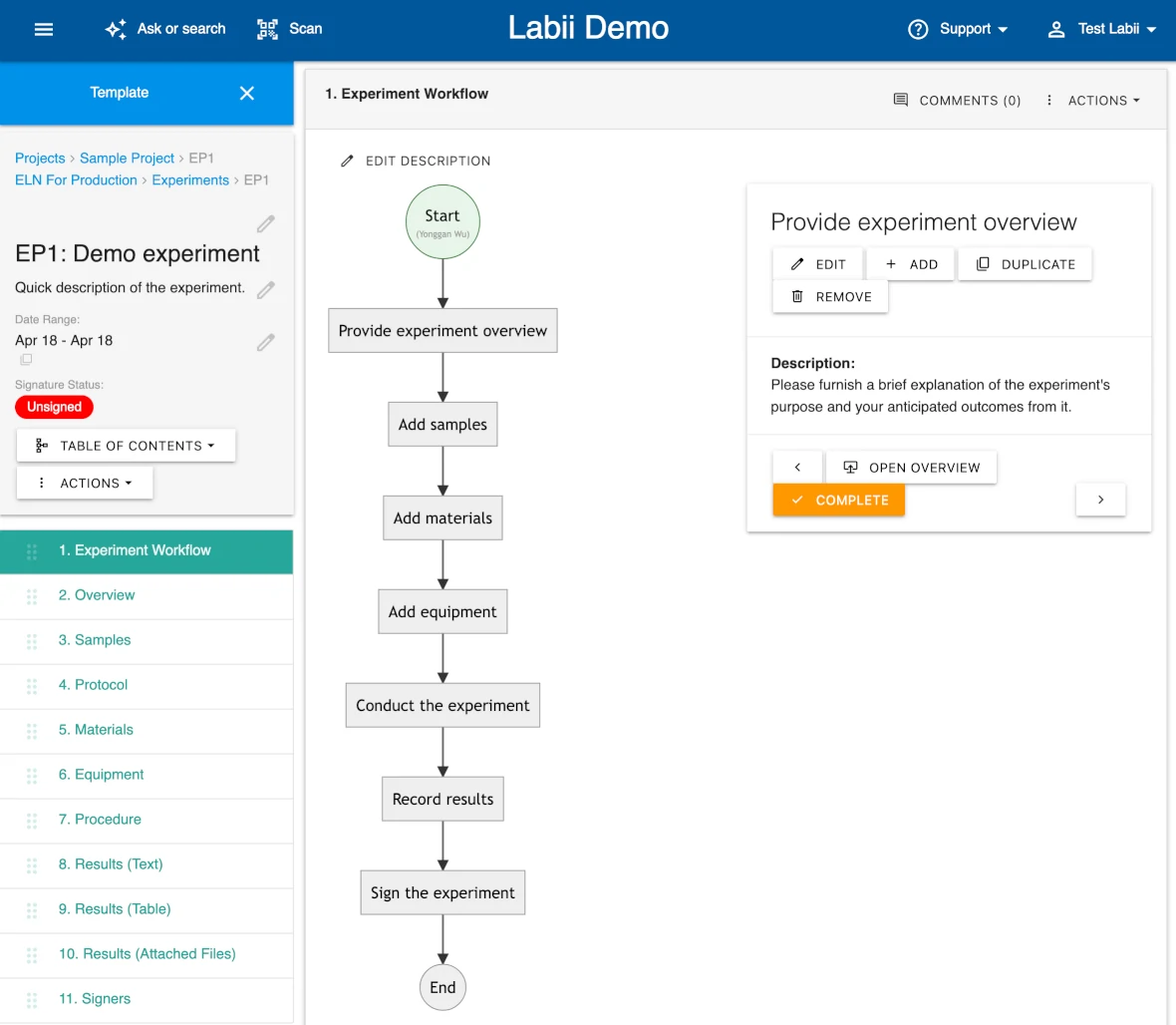
Standardize and Automate Repetitive Workflows

Labii ELN for Production is designed specifically for labs conducting routine and standardized experiments. By providing a framework to document every step with precision—SOPs, samples, equipment, and result fields—Labii ensures repeatability, consistency, and compliance across production runs.
Start with Structured Layouts—No Need to Build from Scratch
Save time and ensure accuracy by using pre-configured experiment templates. Define sections for protocols, materials, instruments, and data capture once, then reuse them across similar experiments without manual setup.
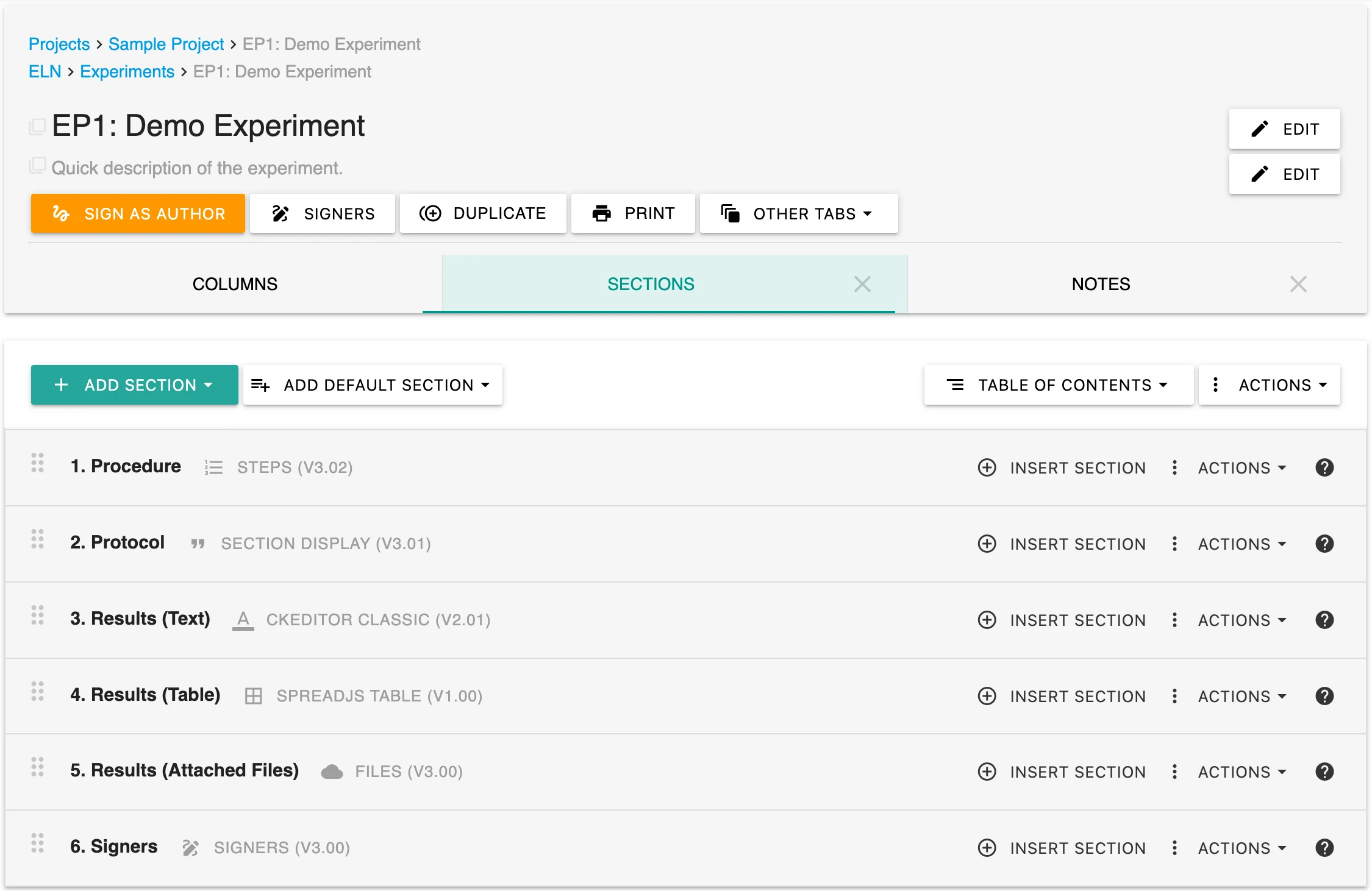
Import SOPs and Prior Data Instantly
Labii’s data loading feature allows users to automatically pull in SOPs, equipment lists, and sample information—either from templates or previous records. This eliminates redundant data entry and improves accuracy and speed when creating new entries.
Build and Execute Multi-Step Experimental Pipelines
Design multi-phase workflows that reflect your lab’s real-world procedures. With support for branching logic, linked steps, and shared resources, Labii helps enforce correct execution while capturing detailed records of each stage.
read_moreLearn more about process integration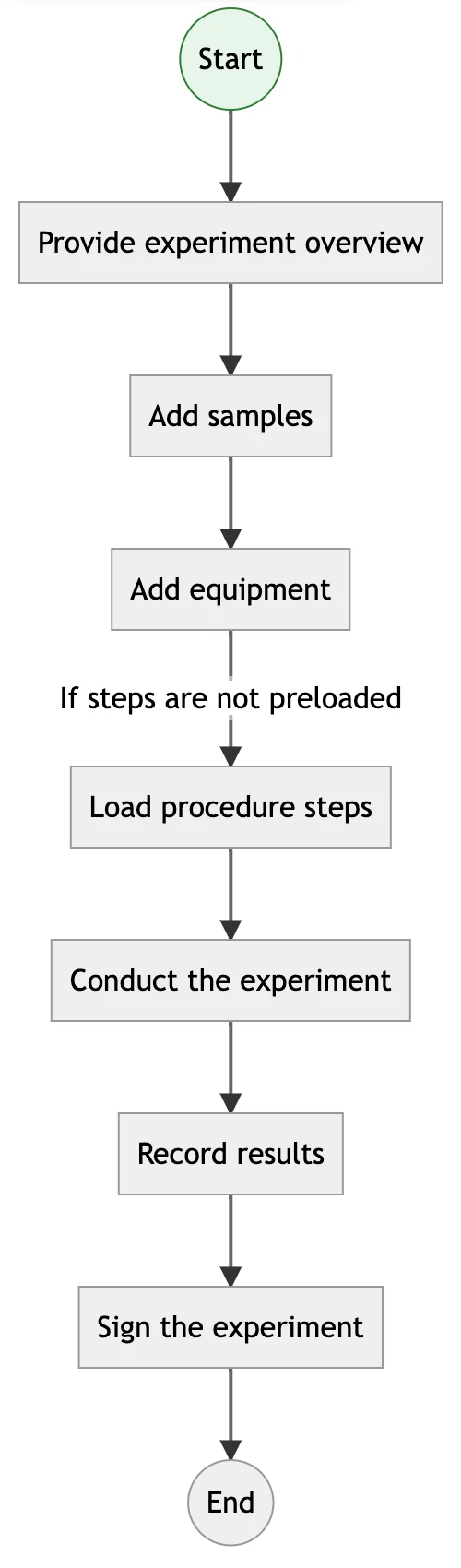
Labii Widgets: Your Solution for Customized Data Display in ELN
Labii ELN serves various scientific disciplines, offers widgets as compact, specialized tools. These widgets integrate seamlessly with columns and sections, enabling efficient data display and collection. Users can select the ideal widget for each section, tailoring data management and visualization to their specific research needs, whether they are biologists, chemists, or professionals from other fields.
Document Every Detail to Meet Industry Standards
Labii’s structured data capture ensures all steps are thoroughly documented to support internal quality control and meet external regulatory standards. Every action is traceable, time-stamped, and securely stored for audit readiness.
Focus on Work, Not Paperwork
By automating data capture and reducing manual entry, Labii dramatically speeds up the documentation process. Teams can spend more time running experiments and less time formatting entries or rewriting procedures.
Tailor Every Section to Fit Your Production Protocols
Whether you follow GLP, ISO, or internal procedures, Labii lets you configure every aspect—fields, sections, workflows, and permissions—so your ELN reflects exactly how your lab works.

Built for Scale, Backed by Real-World Use

Labii ELN for Production is used by research organizations and production labs that demand speed, accuracy, and standardization. Whether managing a few experiments or thousands, Labii is built to grow with your operation.
read_moreLearn more about Digester Doc Story in using Labii ELNReady to Build Your Own Scientific Applications?
See how Labii can transform your research management. Schedule a live demo or start configuring your system today. Whether you’re looking for a complete ELN/LIMS replacement or building your own lab workflows, Labii gives you everything you need to start fast and scale smart.
Refer a company to Labii and earn free months for each seat they sign.